Potassium hydroxide hazards
Home » chemical » Potassium hydroxide hazards >Potassium hydroxide hazards
Potassium Hydroxide Hazards. Fehlings solution contains sodium hydroxide of this concentration. It can cause severe tissue damage and may be fatal if swallowed. Serged and bound seams are degraded by some hazardous liquid chemicals such as strong acids and should not be. Causes severe eye burns.
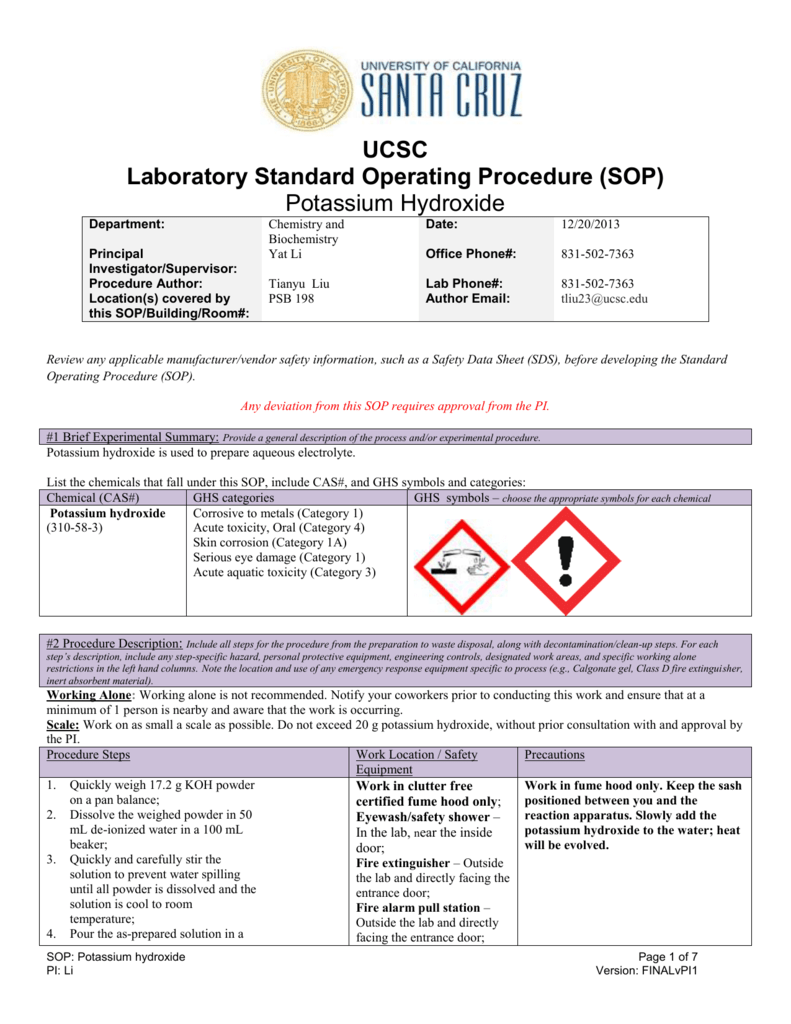 Potassium Hydroxide From studylib.net
Potassium Hydroxide From studylib.net
Lye is extremely corrosive and can burn your skin and eyes. Potassium hydroxide is also a precursor to other potassium compounds. Do not breathe. It can cause severe tissue damage and may be fatal if swallowed. Aim for an overall healthful dietary pattern the American Heart Association advises rather than focusing on good or bad foods. Causes severe digestive and respiratory tract burns.
Lye is extremely corrosive and can burn your skin and eyes.
Potassium hydroxide is corrosive to tissues. What is potassium. The health hazards of potassium hydroxide are similar to those of the other strong alkalies such as sodium hydroxide. Like Mg 2 and Ca 2 ions Al 3 in most situations seeks out complexing agents with oxygen-atom donor sites such as carboxylate and phosphate groups including in biological systems. Potassium hydroxide is an inorganic compound with the formula K OH and is commonly called caustic potash. Respiratory system eyes skin.
 Source: studylib.net
Source: studylib.net
Personal Health A Heart-Healthy Way to Eat. White or yellow solid. Potassium hydroxide is an inorganic compound with the formula K OH and is commonly called caustic potash. Potash lye and its solution can severely irritate skin mucous membranes and eyes. Section 3 - Hazards Identification EMERGENCY OVERVIEW.
 Source: yumpu.com
Source: yumpu.com
When it comes in contact with water or moisture it can generate heat to instigate combustion. Aim for an overall healthful dietary pattern the American Heart Association advises rather than focusing on good or bad foods. Fehlings solution contains sodium hydroxide of this concentration. Potassium hydroxide if less than 04 M but 01 M or more IRRITANT. Respiratory system eyes skin.
 Source: fishersci.com
Source: fishersci.com
Potassium carbonate K2CO3 is a white salt soluble in water insoluble in ethanol which forms a strongly alkaline solutionIt can be made as the product of potassium hydroxides absorbent reaction with carbon dioxideIt presents a large capacity to absorb moisture. Serged and bound seams are degraded by some hazardous liquid chemicals such as strong acids and should not be. Potassium hydroxide soaps are softer and more easily dissolved in water than sodium hydroxide soaps. DOES NOT BURN Reasons for Citation f Potassium Hydroxide is on the Right to. Potential Health Effects Eye.
 Source: www2.atmos.umd.edu
Source: www2.atmos.umd.edu
Liquid drain cleaners contain 25 to 36 of Potassium Hydroxide. Potash lye and its solution can severely irritate skin mucous membranes and eyes. Lye is extremely corrosive and can burn your skin and eyes. Dilute sodium or potassium hydroxide solution Sodium hydroxide If less than 05 M but 0125 M or more. It is used in the home as an oven cleaner.
 Source: yumpu.com
Source: yumpu.com
Potassium carbonate K2CO3 is a white salt soluble in water insoluble in ethanol which forms a strongly alkaline solutionIt can be made as the product of potassium hydroxides absorbent reaction with carbon dioxideIt presents a large capacity to absorb moisture. Causes severe eye and skin burns. Liquid drain cleaners contain 25 to 36 of Potassium Hydroxide. Serged and bound seams are degraded by some hazardous liquid chemicals such as strong acids and should not be. Irritating to the eyes and skin.
 Source: studylib.net
Source: studylib.net
Potassium hydroxide is an inorganic compound with the formula K OH and is commonly called caustic potash. Potassium Hydroxide is an odorless white or slightly yellow flakey or lumpy solid which is often in a water solution. Frequently Asked Questions FAQs. Potassium hydroxide is also a precursor to other potassium compounds. Causes severe eye burns.

What is potassium. White or yellow solid. Potential Health Effects Eye. It is used in making soap as an electrolyte in alkaline batteries and in electroplating lithography and paint and varnish removers. Liquid drain cleaners contain 25 to 36 of Potassium Hydroxide.
 Source: tenaquip.com
Source: tenaquip.com
Respiratory system eyes skin. Potassium hydroxide KOH 1000. Causes severe eye and skin burns. What is potassium. Lye is added to water cooled for a few minutes and then added to oils and butters.

Causes severe digestive and respiratory tract burns. Respiratory system eyes skin. The Hazard fields include special hazard alerts air and water reactions fire hazards health hazards a reactivity profile and. Personal Health A Heart-Healthy Way to Eat. Liquid 480 480 480 480 480 480 480 480 480 480 indicates greater than.

DOES NOT BURN Reasons for Citation f Potassium Hydroxide is on the Right to. Potassium carbonate K2CO3 is a white salt soluble in water insoluble in ethanol which forms a strongly alkaline solutionIt can be made as the product of potassium hydroxides absorbent reaction with carbon dioxideIt presents a large capacity to absorb moisture. Potassium hydroxide also known as lye is an inorganic compound with the chemical formula KOHAlso commonly referred to as caustic potash it is a potent base that is marketed in several forms including pellets flakes and powdersIt is used in various chemical industrial and manufacturing applications. Along with sodium hydroxide NaOH KOH is a prototypical strong baseIt has many industrial and niche applications most of which exploit its caustic nature and its reactivity toward acidsAn estimated 700000 to 800000 tonnes were produced in 2005. Frequently Asked Questions FAQs.
If you find this site good, please support us by sharing this posts to your preference social media accounts like Facebook, Instagram and so on or you can also save this blog page with the title potassium hydroxide hazards by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.