Ethyl acetate hazards
Home » chemical » Ethyl acetate hazards >Ethyl acetate hazards
Ethyl Acetate Hazards. It may also ignite with potassium tert-butoxide. Linking to a non-federal website does not constitute an endorsement by CDC or any of its employees of the sponsors or the information and products presented on the website. AquaPhoenix Scientific 9 Barnhart. Ethyl acetate CH3COOC2H5 or C4H8O2 CID 8857 - structure chemical names physical and chemical properties classification patents literature biological.

Ethyl acetate systematically ethyl ethanoate commonly abbreviated EtOAc ETAC or EA is the organic compound with the formula CH 3 COOCH 2 CH 3 simplified to C 4 H 8 O 2. S25305 Recommended uses of the product and uses restrictions on use. Excerpt from ERG Guide 127 Flammable Liquids Water-Miscible. It is incompatible with nitrates strong alkalis and strong acids. ETHYL ACETATE page 2 of 6 This Fact Sheet is a summary source of information of all potential and most severe health hazards that may result from exposure. Most vapors are heavier than air.
Duration of exposure concentration of the substance and other factors will affect your susceptibility to any of the potential effects described below.
Safety hazards of Ethyl Acetate to potentially exposed workers. It is incompatible with nitrates strong alkalis and strong acids. —– HEALTH HAZARD. Hazards identification Classification of the substance or mixture. Vapors may travel to source of ignition and flash back. No smoking Keep container tightly closed Groundbond.
 Source: nugentec.com
Source: nugentec.com
The Centers for Disease Control and Prevention CDC cannot attest to the accuracy of a non-federal website. It is incompatible with nitrates strong alkalis and strong acids. Ethyl acetate is produced commercially from ethyl alcohol and acetic acid which in turn may be produced from natural ingredients or petroleum derivatives. Linking to a non-federal website does not constitute an endorsement by CDC or any of its employees of the sponsors or the information and products presented on the website. —– HEALTH HAZARD.
 Source: manualzz.com
Source: manualzz.com
001 - dihydromyrcenol 005 - benzyl propionate 005 - linalyl acetate 005 - phenethyl alcohol 001 - aldehyde C-14 10 020 - amyl cinnamaldehyde 001 - mahy shiff 005 - naphthyl ethyl ether 001 - aldehyde C-16 10 005 - geraniol an antimicrobial antiseptic and disinfectant that is used also as an. It is incompatible with nitrates strong alkalis and strong acids. Nail salons are mostly small businesses that employ or contract with trained professionals to provide clients with nail services including but not limited to nail filing and polishing artificial nail application and other hand- and foot-care treatments. Nail technicians working in salons across the United States face possible health hazards every day. Ethyl acetate like methylene chloride is very volatile and hence the point made above also applies here.
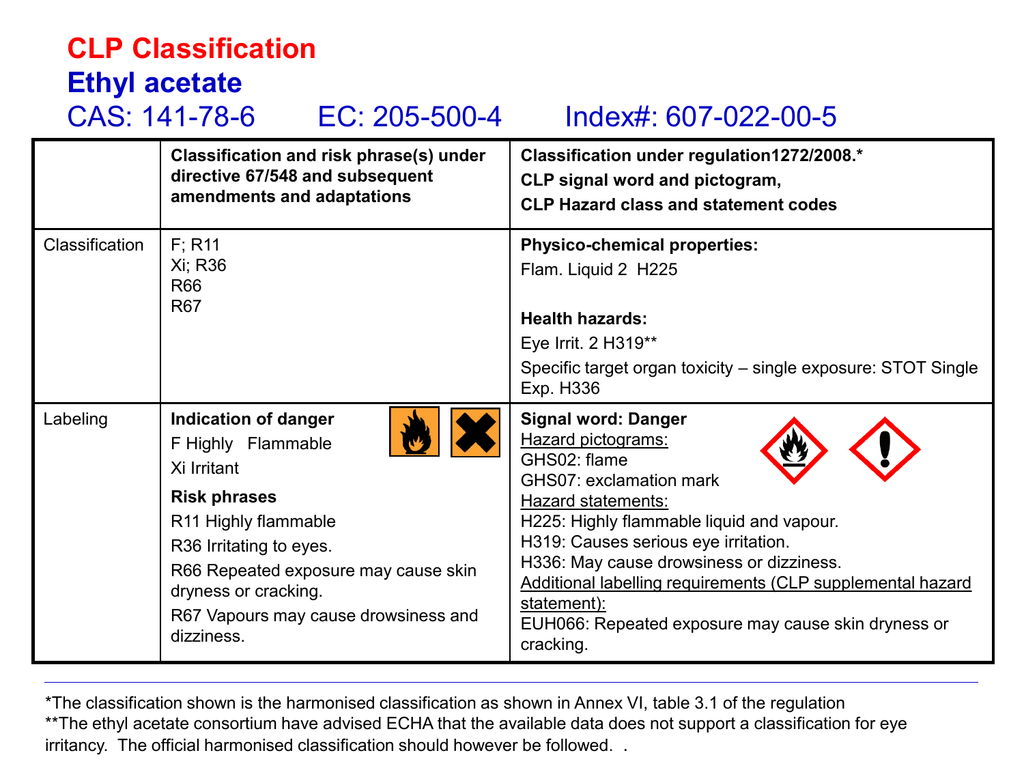 Source: studylib.net
Source: studylib.net
AquaPhoenix Scientific 9 Barnhart. ETHYL ACETATE is also sensitive to heat. Ethyl Acetate Created by Global Safety Management Inc. The Centers for Disease Control and Prevention CDC cannot attest to the accuracy of a non-federal website. Ethyl Acetate ManufacturerSupplier Trade name.
 Source: mysafetylabels.com
Source: mysafetylabels.com
Excerpt from ERG Guide 127 Flammable Liquids Water-Miscible. Now that weve got a basic framework to work with lets have a closer look at the before mentioned methods. Health Hazards Not Otherwise Classified Category 1 Prolonged or repeated contact may dry skin and cause irritation or cracking Flammable liquids _____ Page 1 7 Category 2 _____ Ethyl acetate Revision Date 18-January-2018 Precautionary Statements Prevention Keep away from heat hot surfaces sparks open flames and other ignition sources. No smoking Keep container tightly closed Groundbond. Identification of the substancemixture and of the supplier Product name.
 Source: fishersci.com
Source: fishersci.com
Ethyl Acetate ManufacturerSupplier Trade name. 001 - dihydromyrcenol 005 - benzyl propionate 005 - linalyl acetate 005 - phenethyl alcohol 001 - aldehyde C-14 10 020 - amyl cinnamaldehyde 001 - mahy shiff 005 - naphthyl ethyl ether 001 - aldehyde C-16 10 005 - geraniol an antimicrobial antiseptic and disinfectant that is used also as an. This chemical may ignite or explode with lithium aluminum hydride. Ethyl acetate is produced commercially from ethyl alcohol and acetic acid which in turn may be produced from natural ingredients or petroleum derivatives. This colorless liquid has a characteristic sweet smell similar to pear drops and is used in glues nail polish removers and in the decaffeination process of tea and coffee.

No smoking Keep container tightly closed Groundbond. Ethyl phenylacetate is a volatile aroma component of fruit and honey. Ethyl Acetate ManufacturerSupplier Trade name. Duration of exposure concentration of the substance and other factors will affect your susceptibility to any of the potential effects described below. —– HEALTH HAZARD.
 Source: bradyid.com
Source: bradyid.com
Ethyl Acetate Created by Global Safety Management Inc. Ethyl acetate like methylene chloride is very volatile and hence the point made above also applies here. It will attack some forms of plastics rubber and coatings. Links with this icon indicate that you are leaving the CDC website. It is a constituent of various.

Ethyl acetate CH3COOC2H5 or C4H8O2 CID 8857 - structure chemical names physical and chemical properties classification patents literature biological. Ethyl acetate CH3COOC2H5 or C4H8O2 CID 8857 - structure chemical names physical and chemical properties classification patents literature biological. Ethyl acetate is produced commercially from ethyl alcohol and acetic acid which in turn may be produced from natural ingredients or petroleum derivatives. Identification of the substancemixture and of the supplier Product name. AquaPhoenix Scientific 9 Barnhart.
 Source: studylib.net
Source: studylib.net
Ethyl phenylacetate is a volatile aroma component of fruit and honey. Nail salons are mostly small businesses that employ or contract with trained professionals to provide clients with nail services including but not limited to nail filing and polishing artificial nail application and other hand- and foot-care treatments. Ethyl Acetate Created by Global Safety Management Inc. Ethyl acetate is produced commercially from ethyl alcohol and acetic acid which in turn may be produced from natural ingredients or petroleum derivatives. Will be easily ignited by heat sparks or flames.
 Source: fishersci.com
Source: fishersci.com
Most vapors are heavier than air. Most vapors are heavier than air. Ethyl phenylacetates production and use in food flavorings and as a chemical intermediate may result in its release to the environment through various waste streams. This chemical may ignite or explode with lithium aluminum hydride. Now that weve got a basic framework to work with lets have a closer look at the before mentioned methods.
If you find this site adventageous, please support us by sharing this posts to your own social media accounts like Facebook, Instagram and so on or you can also bookmark this blog page with the title ethyl acetate hazards by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.