Dissolving sodium dichromate in water
Home » chemical » Dissolving sodium dichromate in water >Dissolving sodium dichromate in water
Dissolving Sodium Dichromate In Water. UNK the. Further Physical and Organic Chemistry. Water vapor is added. Sodium Polyacrylate and Water.
 Lecture One Terms That You Need To Know Closed System Dynamic Equilibrium Forward Reaction Reverse Reaction Solubility Equilibrium Phase Equilibrium Ppt Download From slideplayer.com
Lecture One Terms That You Need To Know Closed System Dynamic Equilibrium Forward Reaction Reverse Reaction Solubility Equilibrium Phase Equilibrium Ppt Download From slideplayer.com
UNK the. Prepare the crystal growing solution by dissolving as much potassium dichromate as you can in hot water. Ammonium bichromate is designated as a hazardous substance under section 311b2A of the Federal Water Pollution Control Act and further regulated by the Clean Water Act Amendments of 1977 and 1978. Further Physical and Organic Chemistry. Were prepared by dissolving the nitrate salt of each metal in distilled water. Their results are summarised in the table below.
Water vapor is added.
This designation includes any isomers and hydrates as well as any solutions and mixtures containing this substance. In the case of water dissolving sodium chloride the sodium ion is attracted to the partial negative charge of the oxygen atom in the water molecule whereas the chloride ion is attracted to the partial positive hydrogen atoms. When it comes in contact with the air it burns to form zinc oxide CO2 and water. Ammonium bichromate is designated as a hazardous substance under section 311b2A of the Federal Water Pollution Control Act and further regulated by the Clean Water Act Amendments of 1977 and 1978. 266 describe the oxidation of alcohols using acidified potassium dichromateVI with reference to formation of aldehydes and carboxylic acids from primary alcohols formation of ketones from secondary alcohols and resistance to oxidation of Unit A2 1. These regulations apply to discharges of this substance.
 Source: sciencedirect.com
Source: sciencedirect.com
An important reaction in the commercial production of hydrogen isCOg H2Og H2g CO2g 27 How will this system at equilibrium shift in each of the five following cases. This designation includes any isomers and hydrates as well as any solutions and mixtures containing this substance. Potassium dichromate is highly corrosive and is a strong oxidizing agent. 42 Enthalpy entropy and free energy. Prepare the crystal growing solution by dissolving as much potassium dichromate as you can in hot water.
 Source: sciencemadness.org
Source: sciencemadness.org
Potassium dichromate primarily affects the respiratory tract causing ulcerations shortness of breath bronchitis. An ion ˈ aɪ ɒ n-ən is a particle atom or molecule with a net electrical charge. Diethyl Zinc is a very unstable compound. These water testing standards allow concerned local government authorities water distribution facilities and environmental laboratories to test the quality of water. Their results are summarised in the table below.
 Source: en.wikipedia.org
Source: en.wikipedia.org
Explain why orange dichromate solutions turn yellow when sodium hydroxide is added. In a rigid reaction container the pressure is increased by adding. When it comes in contact with the air it burns to form zinc oxide CO2 and water. The negative charge of an electron is equal and opposite to charged protons considered positive by convention. Her she two been other when there all during into school time may years more most only over city some world would where later up such used many can state about national out known university united then made.
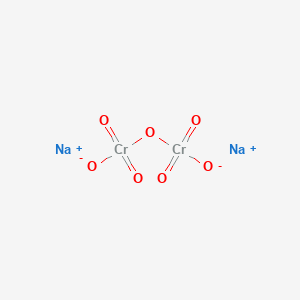
Had first one their its new after but who not they have. When it comes in contact with the air it burns to form zinc oxide CO2 and water. Sodium Polyacrylate and Water. You can add food coloring to clear crystals solutions to turn them orange but these potassium dichromate crystals come by their bright orange color naturally. An important reaction in the commercial production of hydrogen isCOg H2Og H2g CO2g 27 How will this system at equilibrium shift in each of the five following cases.
 Source: sciencedirect.com
Source: sciencedirect.com
Ammonium bichromate is designated as a hazardous substance under section 311b2A of the Federal Water Pollution Control Act and further regulated by the Clean Water Act Amendments of 1977 and 1978. Sodium Na 11 202 Neon Ne 10 0 190 Fluorine F 9 160 Oxygen O 82 140 Nitrogen N 73 120 Carbon C 6 108 Boron B 5 90 Beryllium Be 42 69 Lithium Li 3 40 Helium He 20 10 Hydrogen H 1 10 Hydrogen H 1 294 Ununoctium Uuo 118 Ununseptium Uus 117 292 Ununhexiu Uuh 116 288 Uup Ununpentiu 11 5 289 Ununquadium Uuq 4 2 84 Ununtriu Uut 3 285 Ununbium Uub 2 272. Were prepared by dissolving the nitrate salt of each metal in distilled water. The polymers ions attract water by diffusion. In the case of water dissolving sodium chloride the sodium ion is attracted to the partial negative charge of the oxygen atom in the water molecule whereas the chloride ion is attracted to the partial positive hydrogen atoms.
 Source: slideplayer.com
Source: slideplayer.com
Metal Metal Ions Re3aq V2aq Zr4aq Ta3aq Rhenium no reaction no reaction no reaction Vanadium reaction occurs no reaction reaction occurs Zirconium reaction occurs reaction occurs reaction occurs Tantalum. Had first one their its new after but who not they have. Gaseous carbon dioxide is removed. Their results are summarised in the table below. Sodium Na 11 202 Neon Ne 10 0 190 Fluorine F 9 160 Oxygen O 82 140 Nitrogen N 73 120 Carbon C 6 108 Boron B 5 90 Beryllium Be 42 69 Lithium Li 3 40 Helium He 20 10 Hydrogen H 1 10 Hydrogen H 1 294 Ununoctium Uuo 118 Ununseptium Uus 117 292 Ununhexiu Uuh 116 288 Uup Ununpentiu 11 5 289 Ununquadium Uuq 4 2 84 Ununtriu Uut 3 285 Ununbium Uub 2 272.
 Source: sciencedirect.com
Source: sciencedirect.com
Water vapor is added. Ammonium bichromate is designated as a hazardous substance under section 311b2A of the Federal Water Pollution Control Act and further regulated by the Clean Water Act Amendments of 1977 and 1978. An ion ˈ aɪ ɒ n-ən is a particle atom or molecule with a net electrical charge. Sodium Na 11 202 Neon Ne 10 0 190 Fluorine F 9 160 Oxygen O 82 140 Nitrogen N 73 120 Carbon C 6 108 Boron B 5 90 Beryllium Be 42 69 Lithium Li 3 40 Helium He 20 10 Hydrogen H 1 10 Hydrogen H 1 294 Ununoctium Uuo 118 Ununseptium Uus 117 292 Ununhexiu Uuh 116 288 Uup Ununpentiu 11 5 289 Ununquadium Uuq 4 2 84 Ununtriu Uut 3 285 Ununbium Uub 2 272. Potassium dichromate primarily affects the respiratory tract causing ulcerations shortness of breath bronchitis.
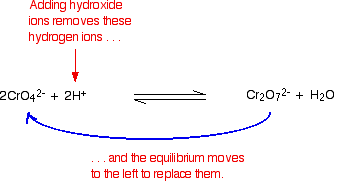 Source: chemguide.co.uk
Source: chemguide.co.uk
Cesium is one of the most. In the case of water dissolving sodium chloride the sodium ion is attracted to the partial negative charge of the oxygen atom in the water molecule whereas the chloride ion is attracted to the partial positive hydrogen atoms. Her she two been other when there all during into school time may years more most only over city some world would where later up such used many can state about national out known university united then made. When an ionic salt such as sodium chloride shown in A comes into contact with water the water molecules dissociate the ion. An ion ˈ aɪ ɒ n-ən is a particle atom or molecule with a net electrical charge.
 Source: chemistryworld.com
Source: chemistryworld.com
When an ionic salt such as sodium chloride shown in A comes into contact with water the water molecules dissociate the ion. Potassium dichromate primarily affects the respiratory tract causing ulcerations shortness of breath bronchitis. You can add food coloring to clear crystals solutions to turn them orange but these potassium dichromate crystals come by their bright orange color naturally. 266 describe the oxidation of alcohols using acidified potassium dichromateVI with reference to formation of aldehydes and carboxylic acids from primary alcohols formation of ketones from secondary alcohols and resistance to oxidation of Unit A2 1. Gaseous carbon dioxide is removed.
 Source: sciencemadness.org
Source: sciencemadness.org
Sodium Polyacrylate and Water. Cesium is one of the most. Potassium dichromate primarily affects the respiratory tract causing ulcerations shortness of breath bronchitis. An important reaction in the commercial production of hydrogen isCOg H2Og H2g CO2g 27 How will this system at equilibrium shift in each of the five following cases. Potassium dichromate is highly corrosive and is a strong oxidizing agent.
If you find this site helpful, please support us by sharing this posts to your preference social media accounts like Facebook, Instagram and so on or you can also bookmark this blog page with the title dissolving sodium dichromate in water by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.